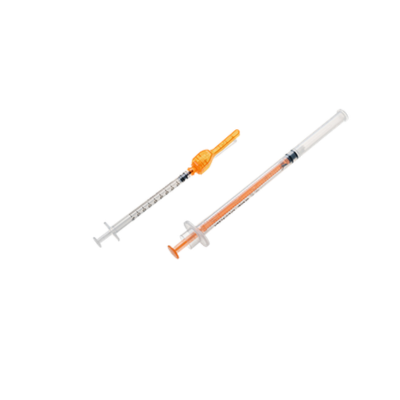
Disposable AV Fistula Needle Line in China
The Disposable AV Fistula Needle Line supports medical-grade material compatibility for needle tubing, hubs, wings, clamps, protectors, and catheter-related components. We can customize feeding tracks, grippers, and contact parts according to your selected polymers, stainless-steel needle specs, and adhesive systems. Pneumatic/electric fittings, valve brands, and connector standards can also be matched to your plant preferences. This helps maintain assembly stability, reduce material mismatch risk, and ensure long-term reliability in high-volume AVF needle manufacturing.
The Disposable AV Fistula Needle Line supports medical-grade material compatibility for needle tubing, hubs, wings, clamps, protectors, and catheter-related components. We can customize feeding tracks, grippers, and contact parts according to your selected polymers, stainless-steel needle specs, and adhesive systems. Pneumatic/electric fittings, valve brands, and connector standards can also be matched to your plant preferences. This helps maintain assembly stability, reduce material mismatch risk, and ensure long-term reliability in high-volume AVF needle manufacturing.
We customize key process modules including automatic assembly, adhesive dispensing, UV/thermal curing, leak-tightness checks, pull-force testing, and visual inspection. Based on your product design (single wing/double wing structures, tubing length, cap type), we optimize cycle time, precision positioning, and bonding parameters. This ensures consistent quality across needle tube, rotating wing, hub, clamp, protector, catheter, and screw cap assemblies. The result is a stable and repeatable automated AVF needle production process.
From semi-automatic stations to fully integrated lines, we provide flexible automation configurations based on budget, labor strategy, and output targets. Options include robotic handling, auto loading/unloading, inline rejection, recipe management, and automatic packaging interfaces. For manufacturers upgrading from manual operations, this modular path reduces transition risk while quickly improving efficiency and product consistency in disposable AV fistula needle manufacturing.
Inspection packages can include CCD vision checks, presence/position verification, bonding quality detection, and barcode/UDI traceability. Digital options include MES connectivity, batch data collection, OEE dashboards, and alarm history records. These functions improve process transparency and support compliance-oriented production management. With real-time monitoring and data-driven optimization, customers can reduce defects, shorten troubleshooting time, and strengthen quality assurance.
We configure line speed, station quantity, buffer design, and packaging connection based on your target annual output and workshop constraints. The modular layout adapts to cleanroom zoning, one-way logistics, and utility access conditions. Whether you need a compact line for pilot production or a high-throughput industrial setup, we provide scalable planning to support phased expansion and faster return on investment.
For regulated production environments, we offer documentation and validation support options such as FAT/SAT protocols, IQ/OQ templates, calibration planning, and process traceability architecture. Quality checkpoints can be configured to your internal SOPs and market requirements. This helps medical manufacturers accelerate qualification and maintain consistent compliance performance during routine production and audits.



Application Fields


Specification for Chronic Renal Failure: Used for arteriovenous fistula puncture in long-term hemodialysis treatment for uremia patients.
Company&Workshop
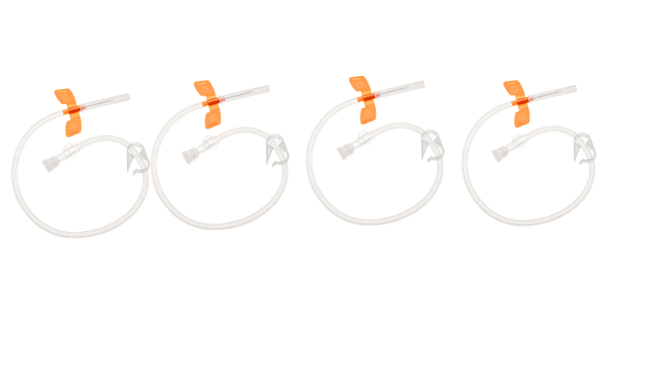

We are a high-tech enterprise specializing in R&D, manufacturing, and sales of hemodialyzer production lines, committed to breaking foreign monopolies and realizing domestic substitution of high-end medical equipment. Our modern standardized workshop adopts clean room design, with precise processing equipment to ensure the stability and precision of hemodialyzer production lines, meeting GMP and ISO 13485 standards.Our professional R&D team cooperates with universities and research institutes, focusing on membrane spinning and automation technology, with annual R&D investment accounting for over 50% of operating income.Corporate Idea Taking “quality is life, innovation drives development” as the core, we provide high-quality, safe, and efficient hemodialyzer production solutions to guard the life health of renal failure patients.
Application Fields
Hemodialysis Consumables Manufacturing
Automated production of disposable AV fistula needles for dialysis treatment.
Large-Scale Medical Device Factories
High-throughput AVF needle assembly for stable mass production.
Manual-to-Automation Upgrades
Replacing manual assembly with intelligent AVF needle production lines.
Quality-Critical Production Environments
Inline inspection and traceability for consistent AVF needle quality.
Cost and Lead-Time Optimization Projects
Improves efficiency, reduces labor intensity, and shortens delivery cycles.
Digital Factory Implementation
MES-ready AVF needle lines with real-time production data integration.
OEM/ODM Medical Consumables Production
Reliable automation for multi-spec disposable AVF needle orders.
Regulated Manufacturing Facilities
Supports validation, documentation, and compliance-oriented operations.